 The U.S. Centers for Disease Control and Prevention (CDC) and the Food and Drug Administration (FDA) are investigating a deadly multistate outbreak of fungal meningitis, which has been linked to steroid injections given for back and joint pain — frequently to older persons suffering from back or joint pain problems.
The U.S. Centers for Disease Control and Prevention (CDC) and the Food and Drug Administration (FDA) are investigating a deadly multistate outbreak of fungal meningitis, which has been linked to steroid injections given for back and joint pain — frequently to older persons suffering from back or joint pain problems.
On October 20, the CDC reported three additional deaths from the outbreak, raising the death toll to 23, and disclosed additional cases of fungal meningitis, increasing the number of recorded cases to 284.
Among the recent cases diagnosed with fungal meningitis that were linked to the tainted steroid were a 53-year-old woman, a 69-year old, and a 71-year-old who received treatment at different Pain Clinics in Florida, news reports have confirmed.
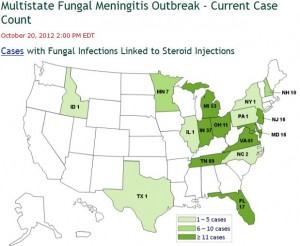
The CDC’s map showing the current case count and the geographic locations of reported cases by state, appears above.
The implicated steroid was manufactured by the New England Compounding Center (NECC) in Framingham, Massachusetts.
“CDC and FDA have confirmed the presence of a fungus known as Exserohilum rostratum in unopened medication vials of preservative-free methylprednisolone acetate (80mg/ml) from one of three implicated lots from NECC (Lot #08102012@51, BUD 2/6/2013),” the FDA and CDC both report.
“The laboratory confirmation further links steroid injections from these lots from NECC to the multistate outbreak of fungal meningitis and joint infections. Testing on the other two implicated lots of methylprednisolone acetate and other NECC injectable medications continues,” the agencies report.
“CDC and state health departments estimate that approximately 14,000 patients may have received injections with medication from three implicated lots of methylprednisolone and nearly 97% of these patients have been contacted for further follow-up,” the FDA states.
NECC has recalled the three affected lots of the streroid. According to the CDC, the affected vials were distributed to healthcare facilities in 23 States. The healthcare facilities have been instructed to notify all affected patients.
“CDC continues to work with states to determine if there may be other fungal infections caused by exposure to NECC products beyond the three lots of preservative-free methylprednisolone acetate (80mg/ml) from NECC that were recalled on September 26, 2012,” the agency advises. The recalled lots include “Lot #05212012@68, BUD 11/17/2012; Lot #06292012@26, BUD 12/26/2012; Lot #08102012@51, BUD 2/6/2013,” according to the CDC.
For a list of healthcare facilities reported by the CDC to have received the affected vials, and a CDC map showing their geographic distribution by state, Click HERE.
Fungal Meningitis and Its Symptoms
The CDC lists the following as symptoms of fungal meningitis:
- New or worsening headache
- Fever
- Sensitivity to light
- Stiff neck
- New weakness or numbness in any part of your body
- Slurred speech
- Increased pain, redness or swelling at your injection site
“If you had an epidural steroid injection since May 21, 2012, and have any of the [above] symptoms, talk to your doctor as soon as possible,” the CDC advises.
What Should Patients Who Have Had Steroid Injections Do?
The CDC Advises:
- “Find out if you received a potentially contaminated medication. If you are concerned about which product was used in your procedure, you should first contact the physician who performed your procedure.
The facilities who received one of the lots recalled on September 26, 2012, are actively contacting patients to find out if they are feeling well. The list of facilities that received medication from one of these three lots is available at http://www.cdc.gov/hai/outbreaks/meningitis-facilities-map.html.
- If you have received a potentially contaminated medication, seek medical attention if you have symptoms. You may have very mild symptoms that are only slightly worse than usual. For example, many infected patients have had slight weakness, slightly worsened back pain, or even a mild headache. Patients with infections have typically developed symptoms within 1-4 weeks after their injection. However, shorter and longer timeframes between injection and onset of symptoms have been reported. The timeframe is still being investigated. You should watch vigilantly for symptoms if you were injected with potentially contaminated steroids and see a doctor if you have any of the symptoms of fungal meningitis or joint infection.”
- If you were contacted because you received a spinal epidural injection with one of the potentially contaminated steroid medications but you feel fine, do you still need to be concerned? “Yes. You should remain vigilant for onset of symptoms because fungal infections can be slow to develop. In this outbreak, symptoms typically have appeared 1 to 4 weeks following injection, but it’s important to know that longer and shorter periods of time between injection and onset of symptoms have been reported. Therefore, you should closely watch for symptoms for at least several months after the injection and see a doctor if you have any of the symptoms below, even if they have been previously evaluated.”
“Patients and clinicians need to remain vigilant for onset of symptoms because fungal infections can be slow to develop, the CDC stresses. “Therefore, patients and physicians need to closely watch for symptoms for at least several months following the injection,” the agency advises.
The CDC suggests that patients see updated Patient Guidance on the CDC’s website for more information, and advises that you contact your physician if you are concerned you may have become ill from your injection.
CDC indicates that it will provide updated guidance as more information becomes available.
Are Patients Who Did Not Receive an Injection At Risk?
No, according to the CDC. “Fungal meningitis is not transmitted from person to person. These infections are associated with a potentially contaminated medication that is injected into the body,” the CDC states.
Thus, only people who received the steroid injections are thought to be at risk, according to the CDC.
More Information
To monitor updates published by the CDC, see the following pages of the CDC Website:
Multistate Fungal Meningitis Outbreak Investigation: Current Situation
Multistate Fungal Meningitis Outbreak – Current Case Count
Frequently Asked Questions for Patients: Multistate Fungal Meningitis Outbreak Investigation
See also the FDA’s Update on Fungal Meningitis.
_____________
Copyright © 2012 Care-Help LLC, publisher of HelpingYouCare®. All rights reserved.



